types: 5. electrons: 10. f orbital. Orbital: shape: flower. types: 7. electrons: 14. Study with Quizlet and memorize flashcards containing terms like 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2, 1s2 2s2 2p6 3s2, 1s2 2s2 2p6 3s2 3p6 4s2 3d5 and more.. I-1 – Iodine (gains 1) 1s2 2s2 2p6 3s2 3p6 – Argon. Write in long term electron configuration and tell which noble gas each is isoelectronic with. Ca+2 – Calcium (loses 2) 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s2 4d10 5p6 – Xenon. Write in long term electron configuration and tell which noble gas each is isoelectronic with.
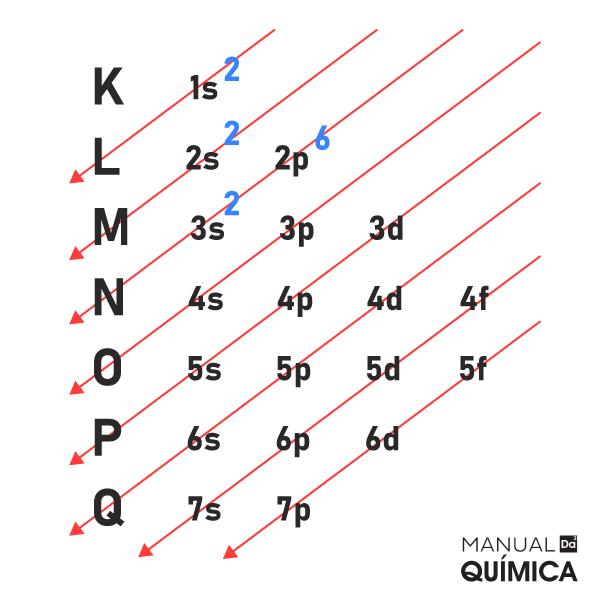
Distribuição eletrônica como fazer e exemplos Manual da Química
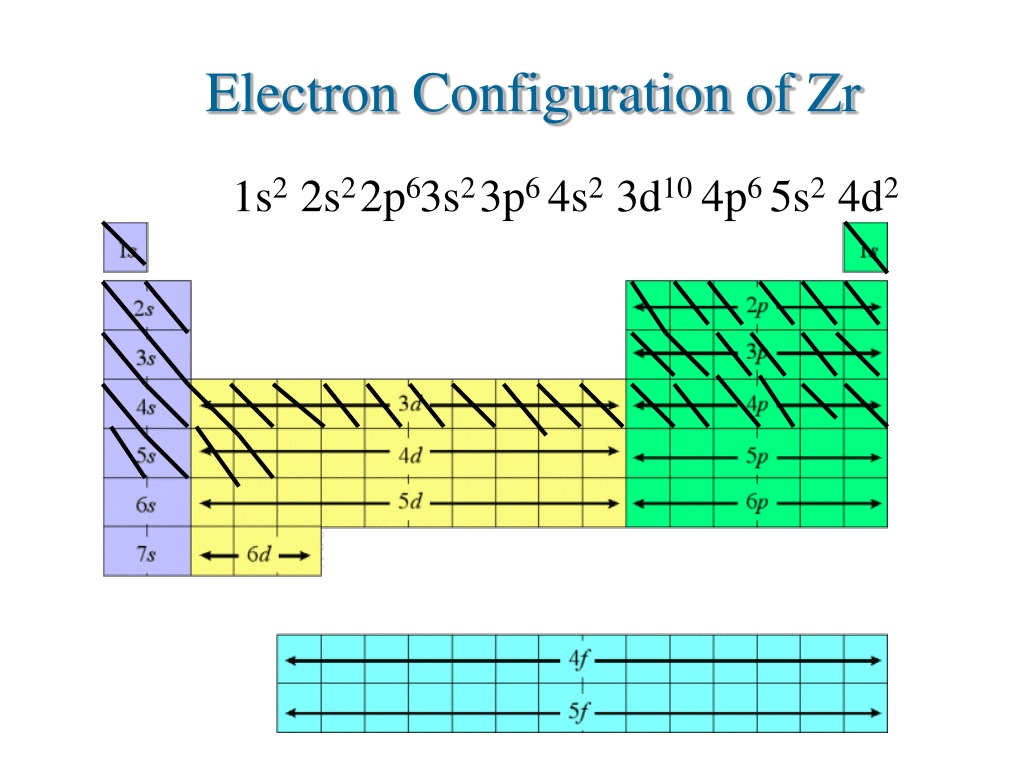
PPT ELECTRON CONFIGURATION PowerPoint Presentation, free download ID9524447

Konfigurasi elektron suatu unsur X adalah 1s2 2s2 2p6 3s2 3p6 4s2 3d10 4p6 5s1 4d5. Unsur
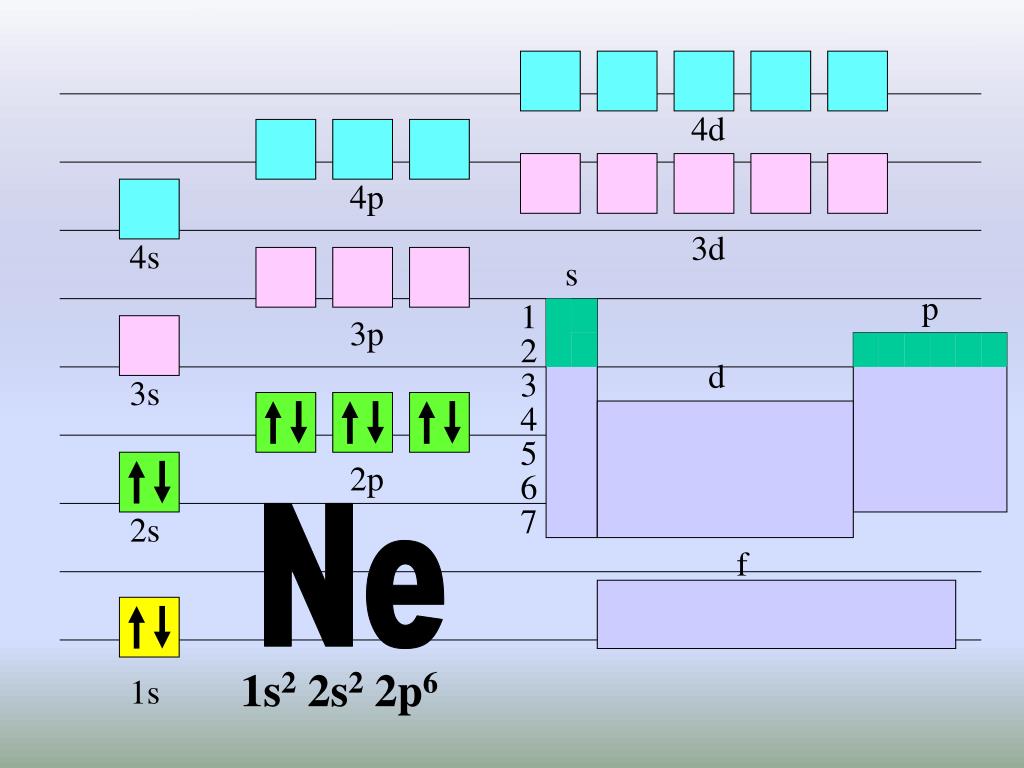
PPT Orbital Diagrams & Electron Configurations PowerPoint Presentation ID4820652

Which element has the electron configuration of? 1s2 2s2 2p6 3s2 3p6 4s1 ? YouTube
Elements Their Atomic, Mass Number,Valency And Electronic Configuratio Periodic Table With

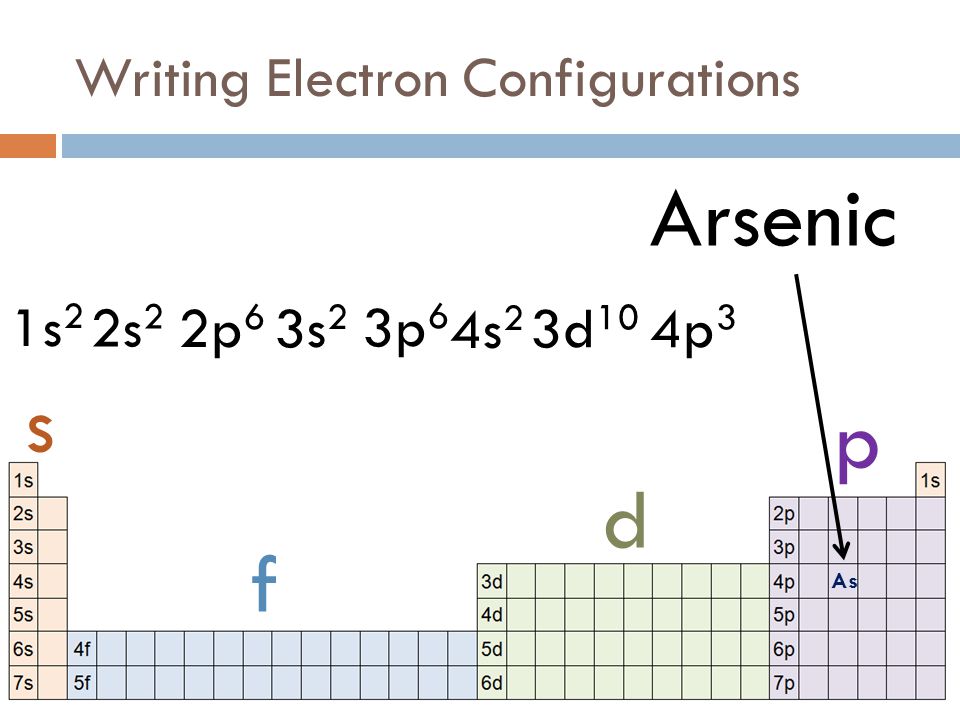
ELECTRON CONFIGURATIONS ppt download

Which element has the electron configuration of 1s2 2s2 2p6 3s2 3p2? YouTube

Which element has the electron configuration of 1s2 2s2 2p6 3s2 3p6 3d1 4s2 YouTube

Which element has the electron configuration of 1s2 2s2 2p6 3s2 3p6 3d10 4s2 4p6 5s2 5p6 YouTube
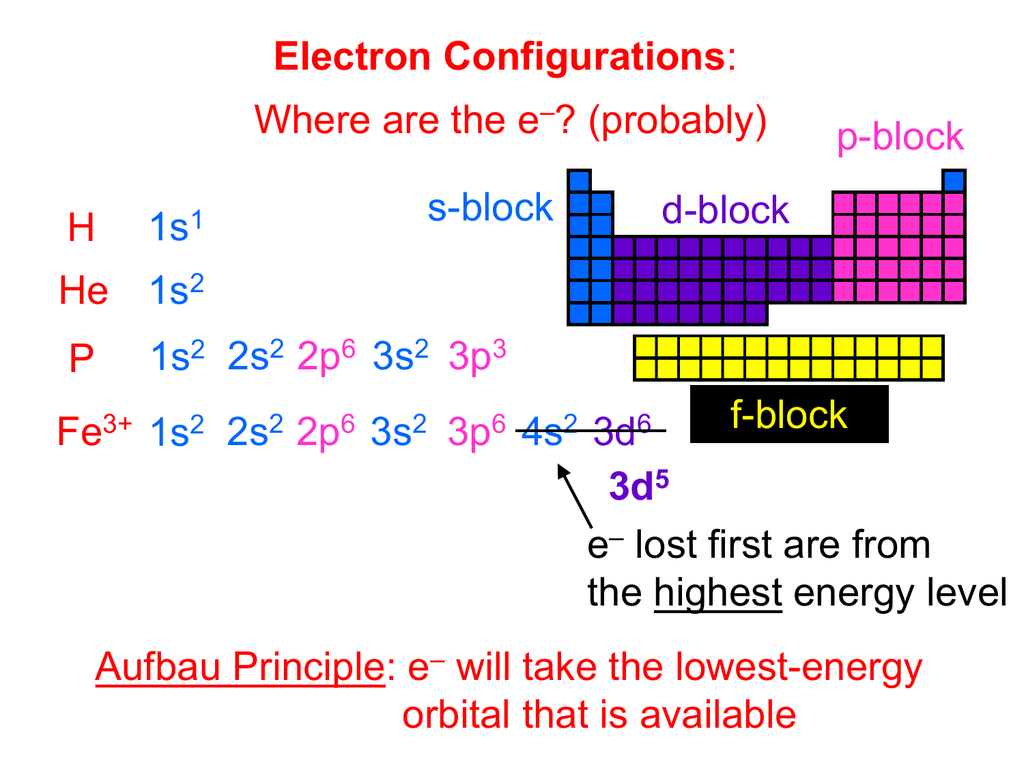
PPT 3

PPT CH 8 Electron Configuration PowerPoint Presentation, free download ID812445

Konfigurasi elektron unsur A dengan nomor atom 28 adalah 1s2 2s2 2p6 3s2 3p6 4s2 3d8 YouTube
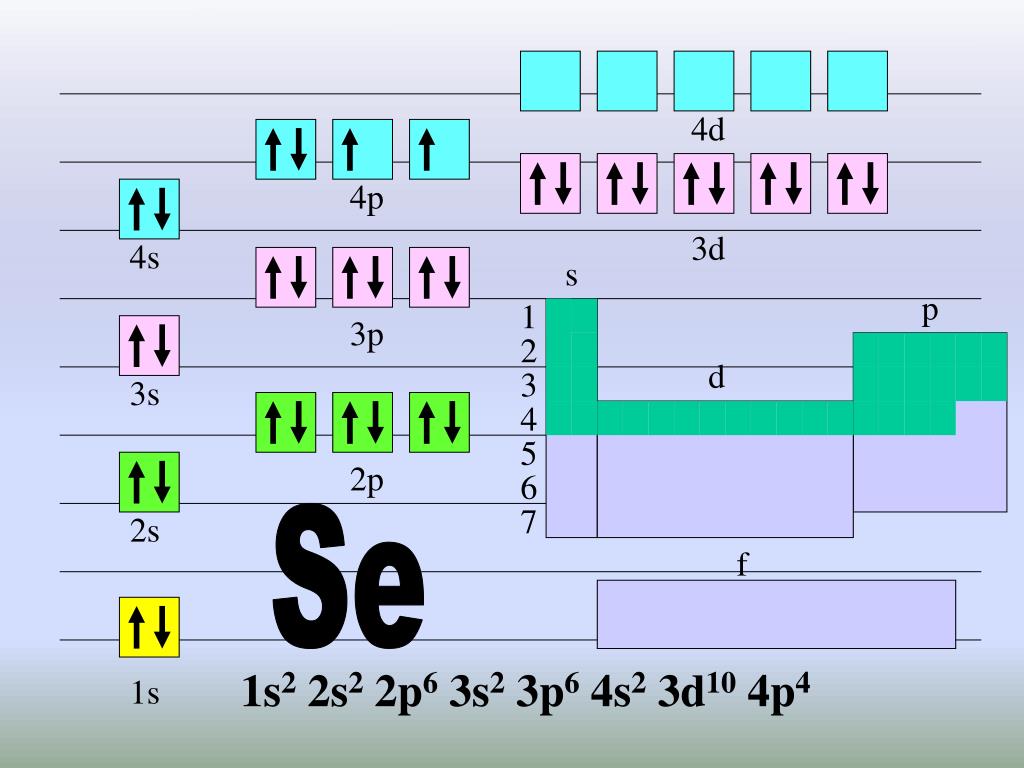
PPT Orbital Diagrams & Electron Configurations PowerPoint Presentation ID4820652

Electron configuration and periodicity online presentation

1s2 2s2 2p6 3s2 3p6 Which element has the electron configuration of 1s2 2s2 2p6 3s2 3p6

PPT And Electron Configurations PowerPoint Presentation, free download ID1873215

PPT Electron Configuration Notes PowerPoint Presentation, free download ID4525941

3.3 electron configuration

Which element has the electron configuration of 1s2 2s2 2p6 3s2 3p6 3d1 4s2 ? YouTube
The full electron configuration of titanium is 1s2 2s2 2p6 3s2 3p6 4s2 3d2 and the abbreviated electron configuration is [Ar]3d24s2. With the Electron configuration of each element, it is possible to specify how the electrons are structured in the atoms of this element. In the case of titanium, its average radius is 140 pm, its covalent radius.. Therefore, one of the 4s2 electrons jumps to the 3d9. This give us the (correct) configuration of: 1s2 2s2 2p6 3s2 3p6 3d10 4s1. For the Cu+ ion we remove one electron from 4s1 leaving us with: 1s 2 2s 2 2p 6 3s 2 3p 6 3d 10. For the Cu2+ ion we remove a total of two electrons (one from the 4s1 and one form the 3d10) leaving us with.




